您好!欢迎访问洛阳富道生物科技有限公司官方网站!
Schwann cells play a crucial role in successful peripheral nerve repair and regeneration by supporting both axonal growth and myelination. Schwann cells are therefore a feasible option for cell therapy treatment of peripheral nerve injury. However, sourcing human Schwann cells at quantities required for development beyond research is challenging. Due to their availability, rapid in vitro expansion, survival, and integration within the host tissue, stem cells have attracted considerable attention as candidate cell therapies. Among them, induced pluripotent stem cells (iPSCs) with the associated prospects for personalized treatment are a promising therapy to take the leap from bench to bedside. In this critical review, we firstly focus on the current knowledge of the Schwann cell phenotype in regard to peripheral nerve injury, including crosstalk with the immune system during peripheral nerve regeneration. Then, we review iPSC to Schwann cell derivation protocols and the results from recent in vitro and in vivo studies. We finally conclude with some prospects for the use of iPSCs in clinical settings.

40层细胞工厂
雪旺氏细胞通过支持轴突生长和髓鞘形成,在成功的周围神经修复和再生中发挥着至关重要的作用。因此,施万细胞是外周神经损伤的细胞疗法治疗的可行选择。然而,以研究之外的开发所需的数量采购人类雪旺氏细胞具有挑战性。由于它们的可用性、快速的体外扩增、存活和在宿主组织内的整合,干细胞作为候选细胞疗法引起了相当多的关注。其中,具有相关个性化治疗前景的诱导多能干细胞 (iPSCs) 是一种很有前景的疗法,可以实现从工作台到床边的飞跃。在这篇批判性评论中,我们首先关注与周围神经损伤有关的雪旺细胞表型的当前知识,包括在周围神经再生过程中与免疫系统的串扰。然后,我们回顾了 iPSC 到 Schwann 细胞衍生协议以及最近体外和体内研究的结果。我们最后总结了在临床环境中使用 iPSC 的一些前景。
Schwann cells play a critical role in peripheral nerve repair through axon guidance and promoting the establishment of a pro-regenerative environment in the nerve bridge. However, primary SCs may not be ideal for efficient use in cell therapies, due mainly to difficulties in purifying and a long expansion process. Despite extensive efforts to develop reliable methods to differentiate stem cells to SCs as an alternative source, both adult stem cells and ESCs have drawbacks—from low purity and yield, non-neuronal differentiation potential, and accessibility of cells, to ethical considerations. Although therapies using differentiated cells sourced from iPSCs will require extensive screening prior to use, iPSCs have the benefits of ESCs without the drawback of ethical concerns which have been cited as a potential barrier to the application of ESC therapies worldwide.

细胞转瓶2L
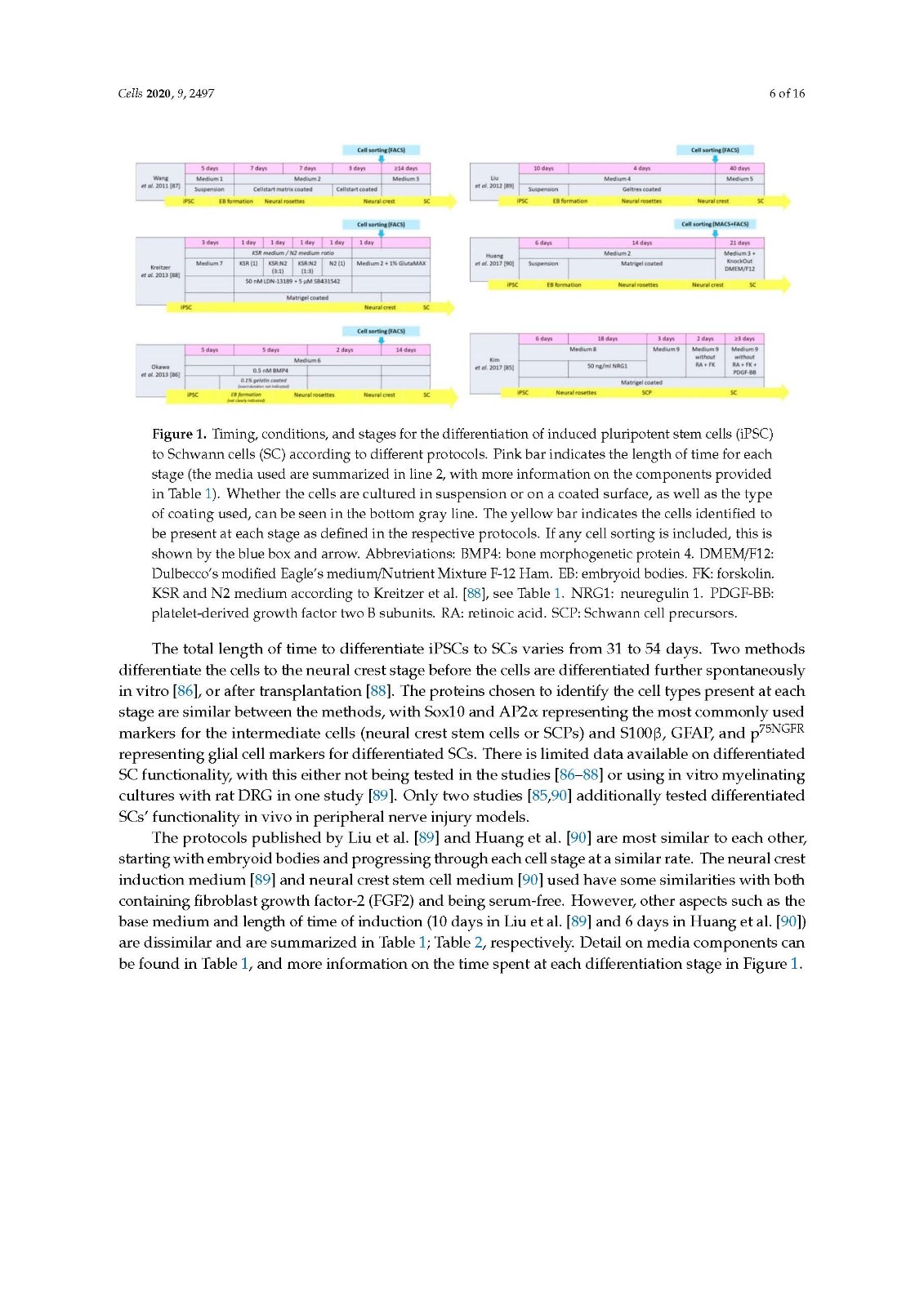
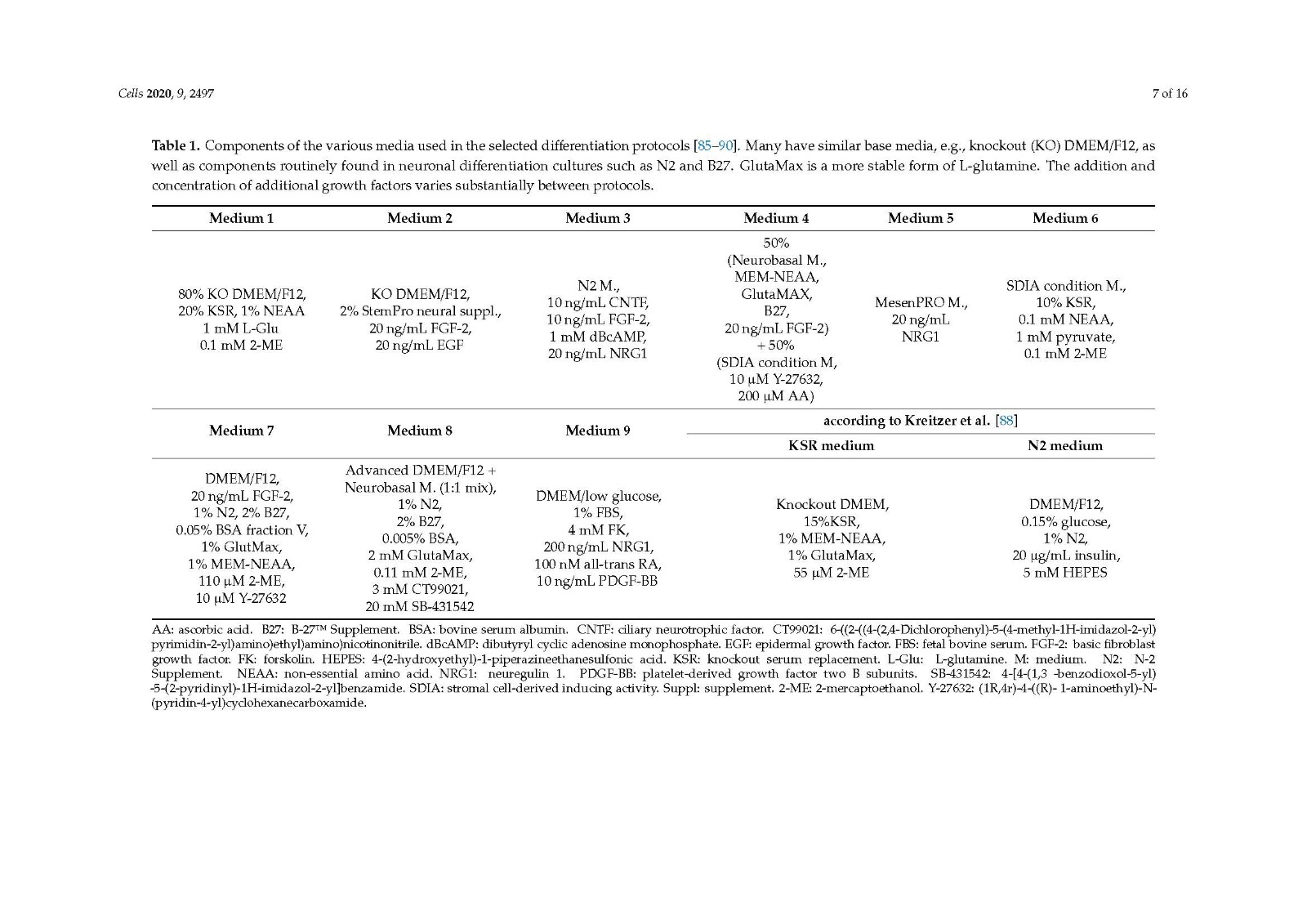
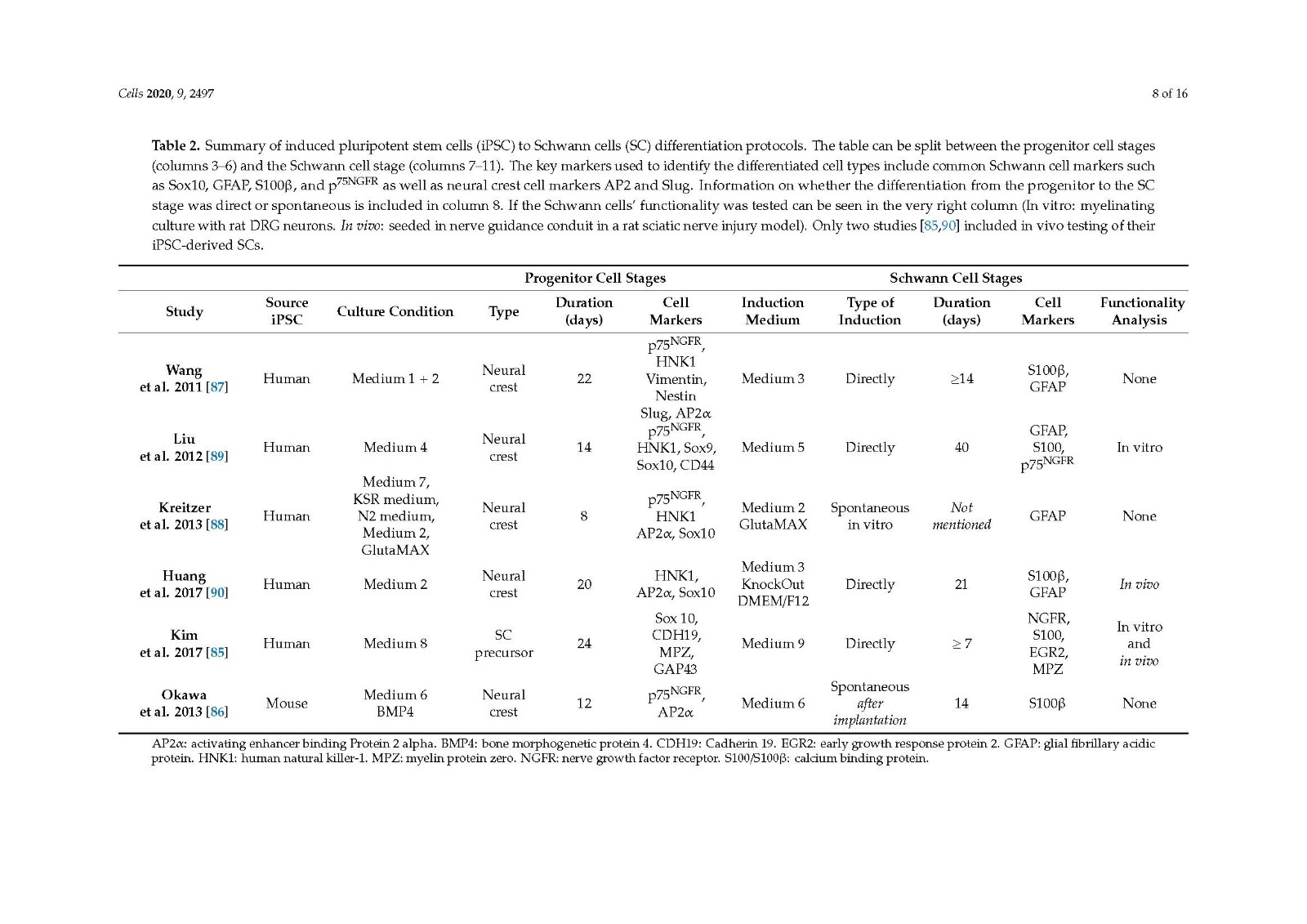
There are several protocols differentiating iPSCs to neural crest stem cells, with the three studies looked at in the current review [85,89,90] including a step to differentiate progenitor cells to SCs, showing promising outcomes both in vitro and in vivo. All three methods generate differentiated cells that express SC markers and release neurotrophic factors. A clear comparison of in vivo outcomes on peripheral nerve repair cannot be made due to the varied choice of animal models used.
It would be beneficial to compare the cell types derived from the different protocols in similar animal models of peripheral nerve injury, the true determinant being how well they perform compared to the autograft in a critical length gap and comprehensive functional analysis. Despite this, the use of iPSCs as a source for Schwann cells for use in future peripheral nerve injury repair therapies remains very promising.
雪旺氏细胞通过轴突引导和促进神经桥中促再生环境的建立,在周围神经修复中发挥关键作用。然而,原代 SCs 可能不是有效用于细胞疗法的理想选择,主要是由于纯化困难和漫长的扩增过程。尽管为开发将干细胞分化为 SCs 作为替代来源的可靠方法做出了广泛的努力,但成体干细胞和 ESCs 都存在缺点——从低纯度和产量、非神经元分化潜力和细胞的可及性,到伦理考虑。尽管使用来自 iPSC 的分化细胞进行治疗需要在使用前进行广泛的筛选,有几种协议分化iPSC的神经嵴干细胞,与当前审查,包括一个步骤分化祖细胞的SC,显示出在体外和体内有前途的结果。所有三种方法都会产生表达 SC 标志物并释放神经营养因子的分化细胞。由于使用的动物模型的选择多种多样,因此无法对周围神经修复的体内结果进行明确的比较。

高效摇瓶5L
在类似的周围神经损伤动物模型中比较来自不同方案的细胞类型将是有益的,真正的决定因素是它们在临界长度间隙和综合功能分析中与自体移植相比的表现如何。尽管如此,使用 iPSC 作为雪旺细胞的来源,用于未来的周围神经损伤修复疗法仍然非常有希望。













来 源:MDPI https://www.mdpi.com/2073-4409/9/11/2497/htm
关键词 雪旺细胞,chwann cells,induced pluripotent stem cells, peripheral nerve,regenerative medicine,诱导多能干细胞,周围神经,再生医学
上一篇: 为什么血清瓶要求无内毒素
下一篇: 细胞转瓶中的细胞贴壁情况差是什么原因