您好!欢迎访问洛阳富道生物科技有限公司官方网站!

10层细胞工厂
Human skin keratinocyte primary cultures can be established from skin biopsies with culture media containing epithelial growth factor (EGF). Although current methods are efficient, optimization is required to accelerate the procedure and obtain these cultures in less time. In the present study, we evaluated the effect of novel formulations based on EGF-loaded nanostructured lipid carriers (NLC). First, biosafety of NLC containing recombinant human EGF (NLC-rhEGF) was verified in immortalized skin keratinocytes and cornea epithelial cells, and in two epithelial cancer cell lines, by quantifying free DNA released to the culture medium. Then we established primary cell cultures of human skin keratinocytes with basal culture media (BM) and BM supplemented with NLC-rhEGF, liquid EGF (L-rhEGF), or NLC alone (NLC-blank). The results showed that cells isolated by enzymatic digestion and cultured with or without a feeder layer had a similar growth rate regardless of the medium used. However, the explant technique showed higher efficiency when NLC-rhEGF culture medium was used, compared to BM, L-rhEGF, or NLC-blank. Gene expression analysis showed that NLC-rhEGF was able to increase EGFR gene expression, along with that of other genes related to cytokeratins, cell–cell junctions, and keratinocyte maturation and differentiation. In summary, these results support the use of NLC-rhEGF to improve the efficiency of explant-based methods in the efficient generation of human keratinocyte primary cell cultures for tissue engineering use.
人类皮肤角质形成细胞原代培养物可以从皮肤活检标本中建立,培养基中含有上皮生长因子 (EGF)。虽然目前的方法是有效的,但需要优化以加快程序并在更短的时间内获得这些文化。在本研究中,我们评估了基于装载 EGF 的纳米结构脂质载体 (NLC) 的新型制剂的效果。首先,通过量化释放到培养基中的游离 DNA,在永生化皮肤角质形成细胞和角膜上皮细胞以及两种上皮癌细胞系中验证了含有重组人 EGF (NLC-rhEGF) 的 NLC 的生物安全性。然后我们用基础培养基 (BM) 和 BM 建立了人类皮肤角质形成细胞的原代细胞培养物,其中补充了 NLC-rhEGF、液体 EGF (L-rhEGF) 或单独的 NLC (NLC-空白)。结果表明,无论使用何种培养基,通过酶消化分离并在有或没有饲养层的情况下培养的细胞都具有相似的生长速率。然而,与 BM、L-rhEGF 或 NLC 空白相比,当使用 NLC-rhEGF 培养基时,外植体技术显示出更高的效率。基因表达分析表明NLC-rhEGF能够增加EGFR基因表达,以及与细胞角蛋白、细胞-细胞连接和角质细胞成熟和分化相关的其他基因的表达。总之,这些结果支持使用 NLC-rhEGF 来提高基于外植体的方法在有效生成用于组织工程的人角质形成细胞原代细胞培养物的效率。
For the biosafety analyses, immortalized human epithelial cells Ker-CT (CRL-4048), and corneal epithelial cells SIRC (ATCC CCL-60) were purchased from ATCC (Rockville, MD, USA) and cultured with the media recommended by the supplier. Cell lines corresponding to epidermoid carcinoma cells A431 (CRL-1555) and lung carcinoma cells A549 (CRM-CCL-185) were also obtained from ATCC and cultured as recommended.
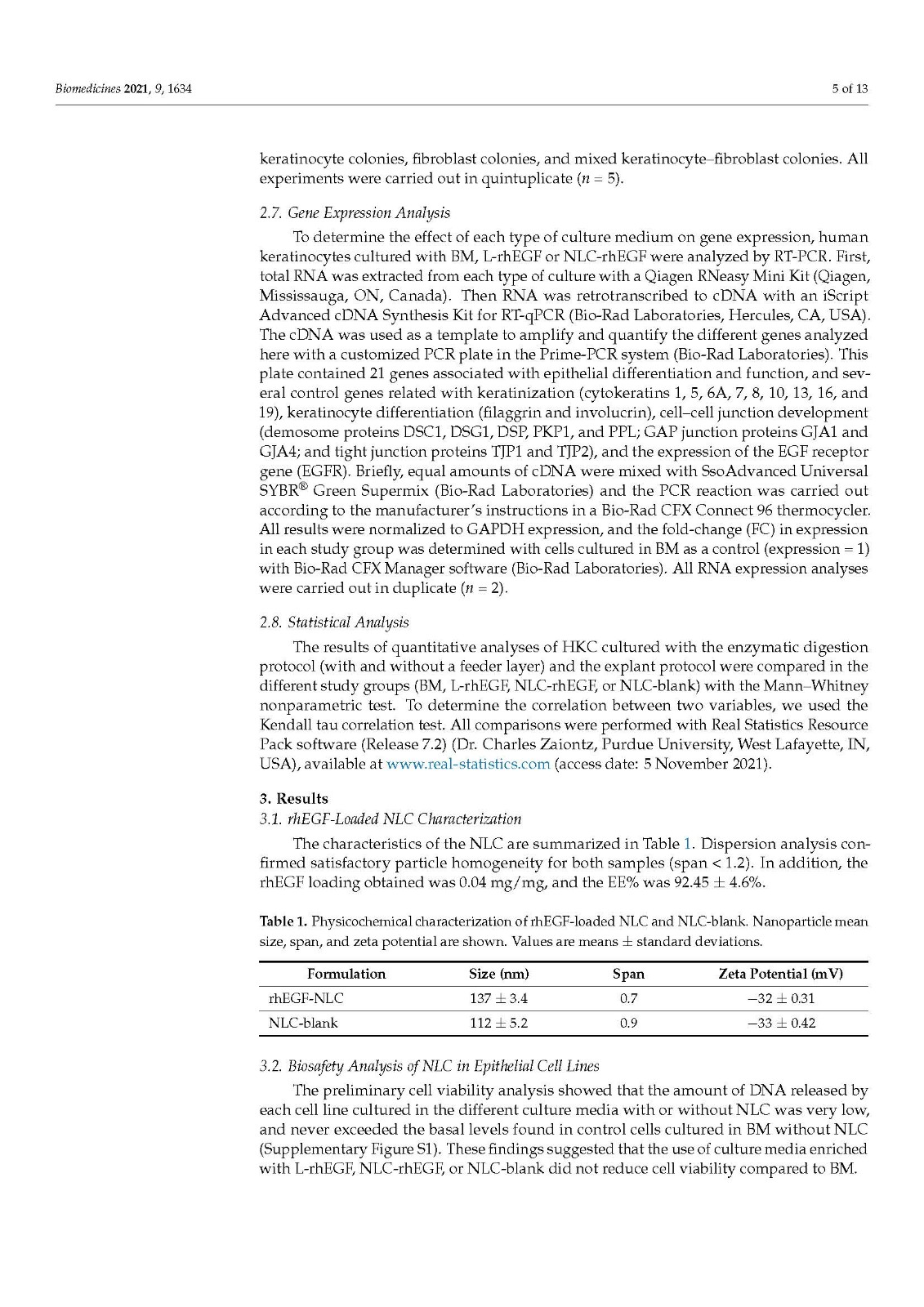
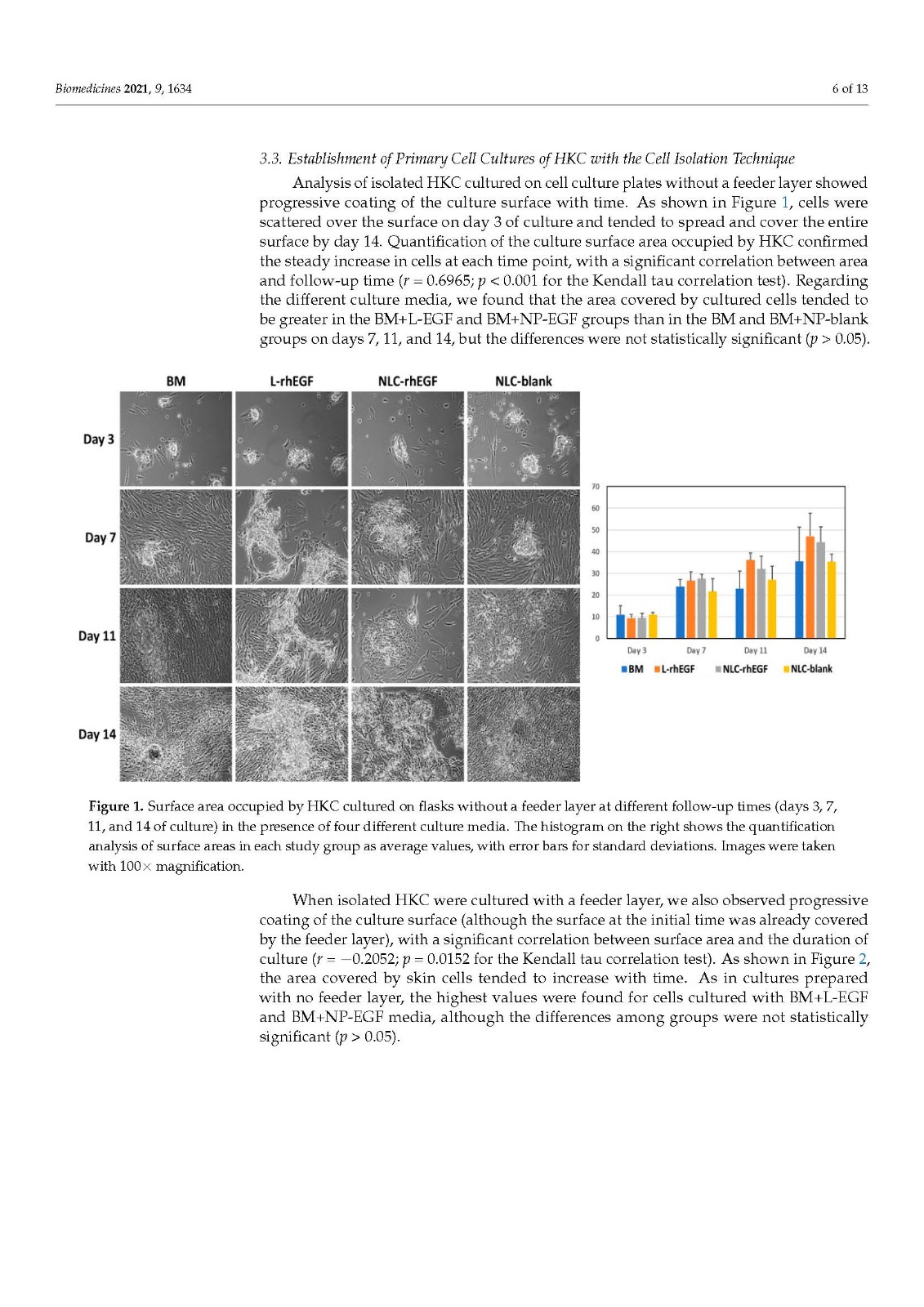
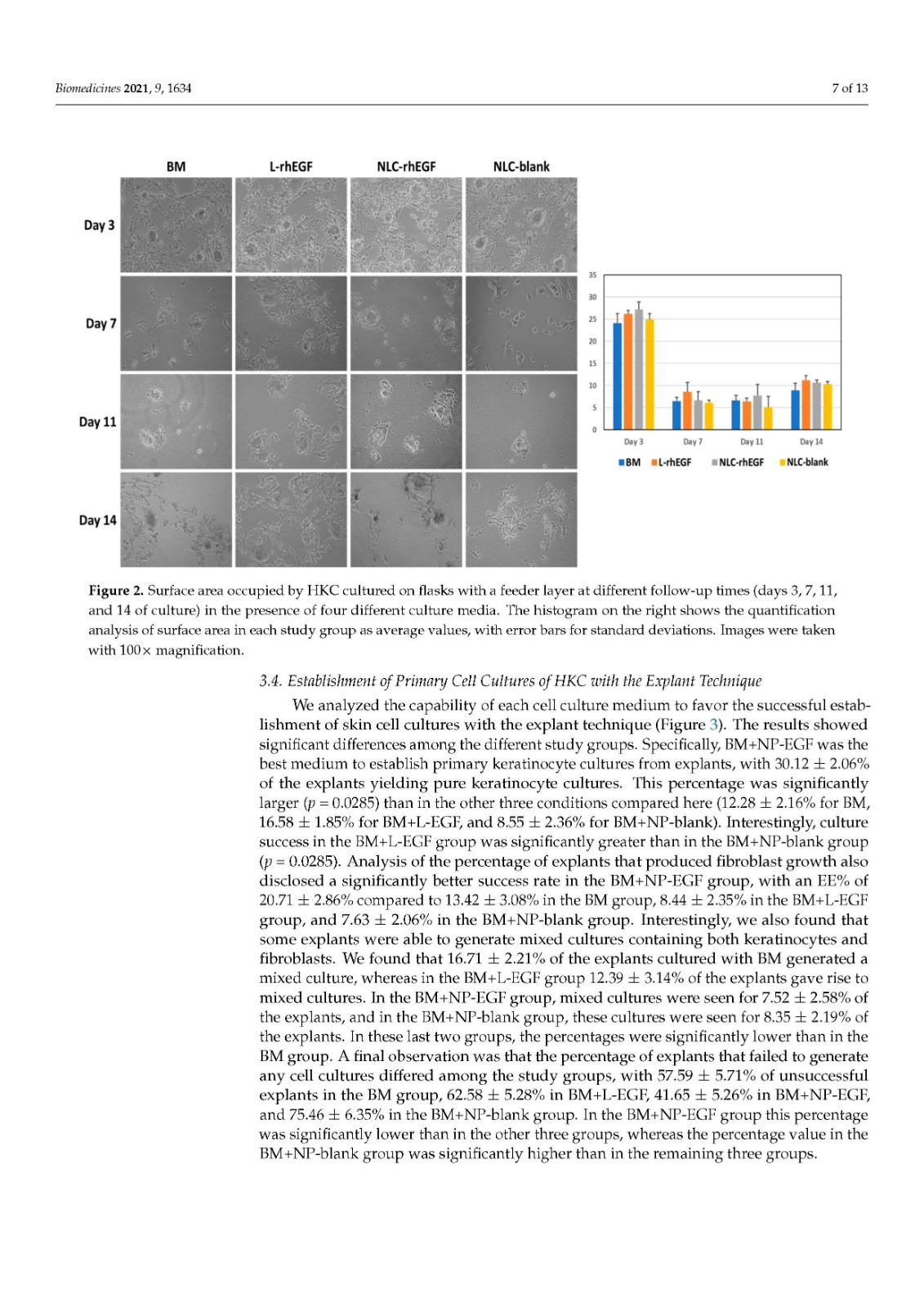
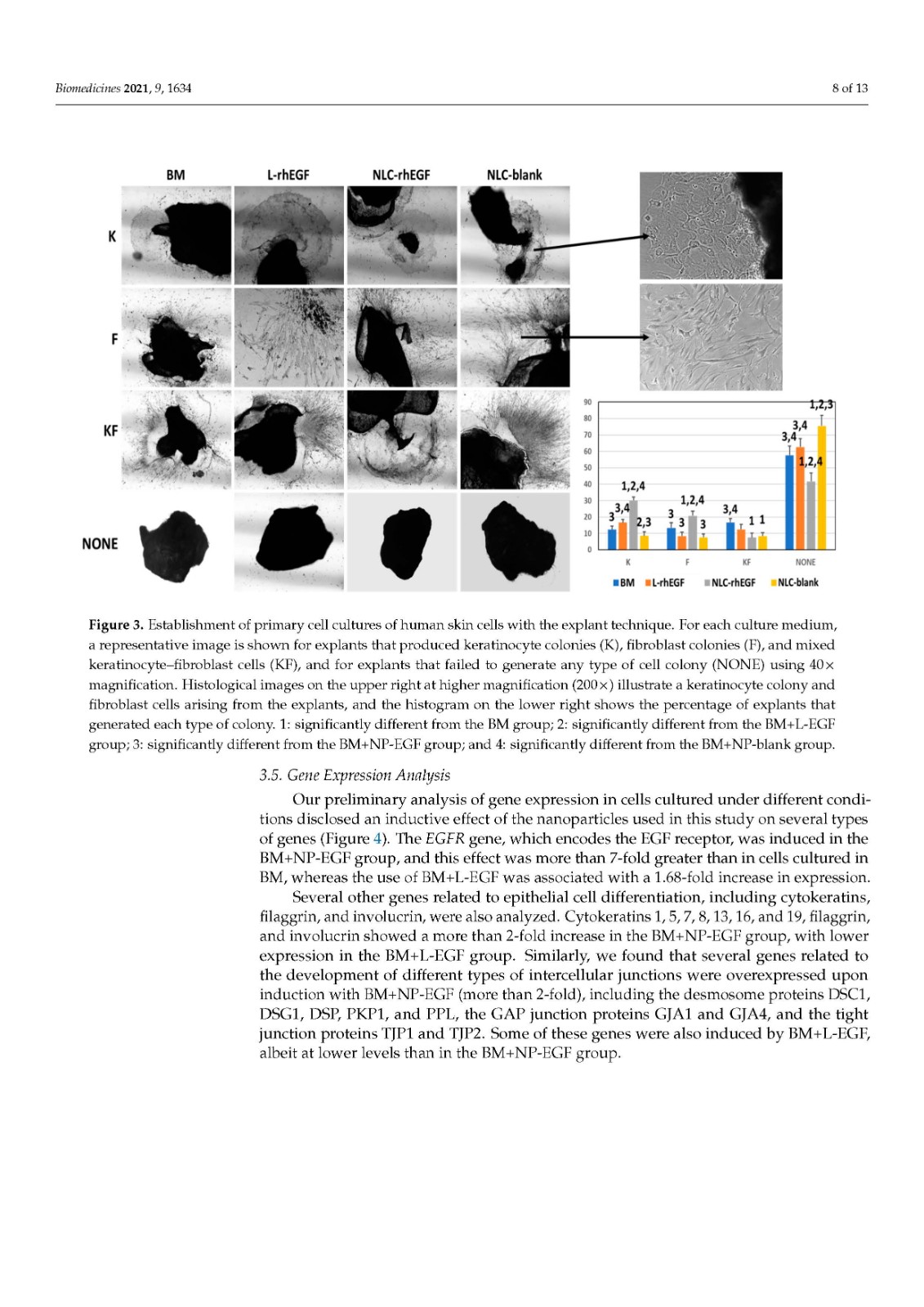
Primary cell cultures of human skin keratinocytes (HKC) were established from small full-thickness skin biopsies obtained from healthy donors who underwent skin surgery. Immediately after excision, skin samples were kept at 4 °C in Dulbecco’s modified Eagle’s medium (DMEM; Merck, Darmstadt, Germany), supplemented with antibiotics and antimycotics (100 U/mL penicillin G, 100 mg/mL streptomycin, and 0.25 mg/mL amphotericin B; Merck), and delivered to the laboratory. Biopsy samples were then washed twice in phosphate-buffered saline (PBS), surgically prepared to remove any remaining adipose tissue, and processed with two different methods routinely used to establish keratinocyte cultures:
(1)
Cell isolation by enzymatic digestion. Tissue samples were incubated in trypsin-EDTA as previously reported [24]. Briefly, samples were placed in a commercial solution containing 0.05% trypsin and 0.02% EDTA (Merck) at 37 °C with gentle shaking. After 20 min, the solution was harvested and inactivated with culture medium, and detached epithelial cells were harvested from the dissociation solution by centrifugation. This procedure was repeated up to 10 times, and all harvested cells were cultured together in 6-well plates. In one of the study groups, harvested cells were cultured on a feeder layer previously cultured on the plates. For this group, 3T3 cells (Merck) were cultured on 6-well plates and a lethal dose of gamma irradiation was applied when cells reached semiconfluence. The cells were washed with PBS, and then dissociated cells were cultured directly on the surface of this cell layer.
(2)
Explant technique. The tissues were trimmed into small pieces with a surgical blade, and each piece was placed on the surface of a 6-well plate, with the epithelial layer in direct contact with the culture surface. These explants were allowed to attach to the surface for 30 min, and a very small amount of culture medium was carefully added to prevent explant detachment. Culture plates were placed in a cell incubator, and 5 mL of culture medium was added to each well 24 h later.
In all cases, cells were incubated at 37 °C with 5% CO2 under standard culture conditions and with specific culture media in each study group (see Section 2.4). The culture medium was changed every 3 days.
细胞培养
对于生物安全分析,永生化人上皮细胞 Ker-CT (CRL-4048) 和角膜上皮细胞 SIRC (ATCC CCL-60) 购自 ATCC (Rockville, MD, USA),并使用供应商推荐的培养基进行培养。对应于表皮样癌细胞 A431 (CRL-1555) 和肺癌细胞 A549 (CRM-CCL-185) 的细胞系也从 ATCC 获得并按推荐培养。
人类皮肤角质形成细胞 (HKC) 的原代细胞培养物是从接受皮肤手术的健康供体获得的小型全层皮肤活检组织中建立的。切除后立即将皮肤样品保存在 Dulbecco 改良 Eagle 培养基(DMEM;默克,德国达姆施塔特)中 4°C,辅以抗生素和抗真菌剂(100 U/mL 青霉素 G、100 mg/mL 链霉素和 0.25 mg/ mL 两性霉素 B;Merck),并送到实验室。然后将活检样本在磷酸盐缓冲盐水 (PBS) 中洗涤两次,通过手术准备去除任何剩余的脂肪组织,并使用两种常规用于建立角质形成细胞培养物的不同方法进行处理:
(1)
通过酶消化分离细胞。如先前报道的那样,组织样品在胰蛋白酶-EDTA 中孵育 [ 24]。简而言之,在 37°C 下,将样品放入含有 0.05% 胰蛋白酶和 0.02% EDTA (Merck) 的商业溶液中,轻轻摇动。20分钟后,收获溶液并用培养基灭活,通过离心从解离溶液中收获分离的上皮细胞。该过程重复多达 10 次,并将所有收获的细胞一起培养在 6 孔板中。在其中一个研究组中,收获的细胞在先前在平板上培养的饲养层上培养。对于该组,3T3 细胞(Merck)在 6 孔板上培养,当细胞达到半融合时应用致死剂量的 γ 辐射。用PBS洗涤细胞,然后将解离的细胞直接培养在该细胞层的表面上。
(2)
外植技术。用手术刀将组织剪成小片,每片置于6孔板表面,上皮层与培养面直接接触。这些外植体被允许附着在表面 30 分钟,并小心地加入非常少量的培养基以防止外植体脱离。将培养板置于细胞培养箱中,24 h后每孔加入5 mL培养基。
在所有情况下,细胞在 37 °C 下与 5% CO 2在标准培养条件下以及每个研究组中的特定培养基一起孵育。每3天更换一次培养基。








关键词:表皮生长因子,纳米粒子,国家图书馆,角质形成细胞,组织工程,细胞培养
EGF, nanoparticles, NLC, keratinocytes, tissue engineering, cell culture
来源:MDPI https://www.mdpi.com/2227-9059/9/11/1634/htm
上一篇: 从血清的储存要求看血清瓶的材质特点
下一篇: 细胞摇瓶培养液体菌种的要点